SPONSOR: Hollister Biosciences Inc. (HOLL:CSE) A vertically integrated Cannabis company with products in 230 California dispensaries, over 80 dispensaries throughout Arizona, joint ventures, licensing agreements and partnerships with Global brands. Revenue generated for the three-month period ended June 30th, 2020 was USD $8.47 million. Learn More

July Cannabis Sales Surge in 5 Western States
- In July 2020, cannabis sales for state licensed retailers and delivery services reached $334.4 million, growing 15 percent from the trailing month and 26 percent compared to July 2019.
- Year to date, sales in California have reached $1.9 billion, a 14 percent increase compared to the same period in 2019.
A Detailed Look at Cannabis Sales in California, Oregon, Arizona, Colorado and Nevada
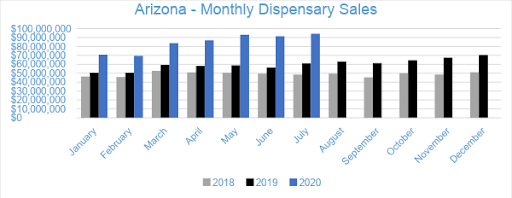
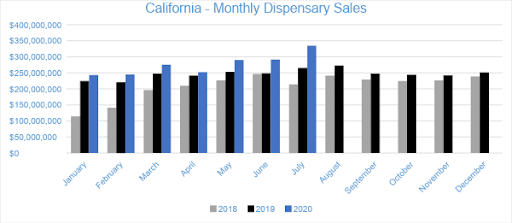
Data provided by BDSA indicates that cannabis sales continued their robust expansion in 5 Western states. During July, the eleventh month after the onset of the vaping crisis and the fifth month of the pandemic impacting the market, sales across the five markets totaled $842.8 million, up 12% from June, which sequential increases of 2% in May and 14% in April. March had spiked to $666.0 million from $579.9 million in February as customers purchased cannabis ahead of store closures and concerns about limitations on access, so clearly cannabis sales are back on track. With the near-term effects of the pandemic now behind us, it’s clear that cannabis demand is surging, even in relatively mature markets, like Oregon and Colorado. Nevada continued to recover as tourism slowly returns, with sales growing 23%. California stood out with 15% sequential growth, while Colorado expanded 13%. Oregon sales climbed 6% and Arizona by 3%, but both of these states had meteoric growth from a year ago. July had one more day than June, which has the effect of adding 3% roughly.
Overall sales growth from a year ago among the five Western markets ranged from 26% in California to as high as 54% in Arizona. Nevada has been impacted by the tourist slowdown but still grew 37% from a year ago. Oregon continued its strong growth, rising 44%, while Colorado grew 35%. Concentrates represented 22-37% of sales by market, below the 26-38% share of sales last August before the vaping crisis hit.
Here is a closer look at each market, as detailed by BDSA:
Arizona
In July 2020, cannabis sales in Arizona’s medical dispensaries reached $94.2 million, growing three percent from June and over 54 percent compared to the previous July. Year-to-date through July, sales have reached $589.2 million, increasing by $194.5 million and 49 percent compared to the same period last year.
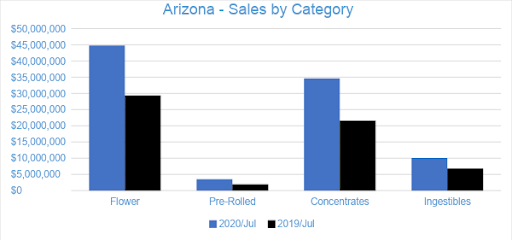
Flower sales accounted for nearly 48 percent of overall revenue for the month with $44.8 million in sales, growing one percent from June. Pre-rolled Joints, which are tracked as a separate category, accounted for just under four percent of revenue with $3.5 million in sales. Compared to the previous July, Flower sales increased by 52 percent and Pre-rolled Joints increased by 83 percent.
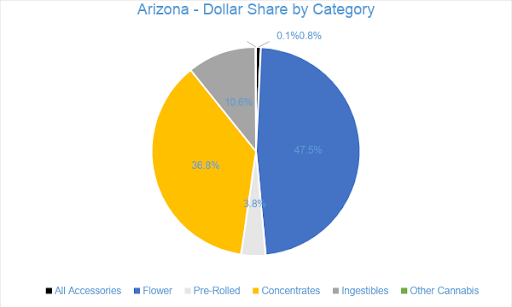
Concentrate sales accounted for about 37 percent of overall revenues with sales totaling $34.6 million in July, increasing six percent from the trailing month. Vape products, the largest segment of the Concentrates category, generated $21.8 million in sales, accounting for 63 percent of the Arizona Concentrates market and over 23 percent of sales across all categories. Compared to July 2019, sales of Dabbable Concentrates grew by 92 percent, while Vape sales grew by 46 percent.
The Ingestibles category accounted for nearly 11 percent of sales in July with $10 million in revenue for the month. Compared to July 2019 and June 2020, sales in the category grew by 47 percent and by four percent, respectively. In July, the $9.3 million in sales from Edibles generated nearly 93 percent of Ingestibles sales overall, while Sublinguals made up the remaining seven percent. Compared to the previous July, Edibles sales increased by 59 percent and Sublingual sales decreased by 30 percent.
California
BDSA sales reporting for the month of July reflects the state market of 699 licensed dispensary locations and 306 delivery operators.
In July 2020, cannabis sales for state licensed retailers and delivery services reached $334.4 million, growing 15 percent from the trailing month and 26 percent compared to July 2019. Year to date, sales in California have reached $1.9 billion, a 14 percent increase compared to the same period in 2019.
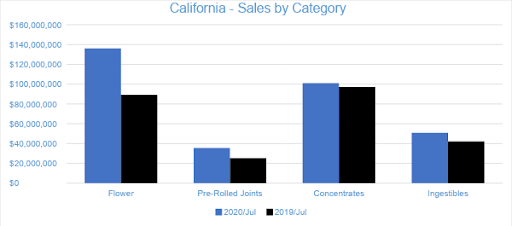
Sales from Flower generated $136.2 million in July 2020, accounting for nearly 41 percent of overall revenues. Compared to the trailing month and July 2019, Flower sales grew by 17 percent and 53 percent, respectively. Pre-rolled Joints, which are tracked as a separate category, accounted for nearly 11 percent of overall July revenues, with $35.6 million in sales.
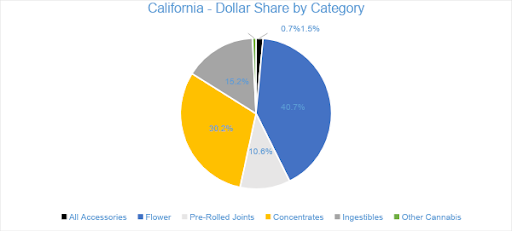
Concentrates were the second-largest revenue category in July 2020, reaching $101 million in sales and accounting for over 30 percent of total revenue. Compared to the trailing month and July 2019, sales increased by 13 percent and by four percent, respectively. The $76 million in Vape sales accounted for over 75 percent of the Concentrates category and 23 percent of the month’s overall sales revenue.
In July 2020, Ingestibles generated $50.8 million in sales, increasing by 11 percent from the trailing month. Compared to July 2019, sales of Ingestibles grew by nearly 21 percent. Edibles contributed 86 percent of Ingestibles revenue, while Sublinguals made up the remaining 14 percent. Compared to July 2019, the Edibles and Sublinguals categories grew by 32 percent and declined by 22 percent, respectively.
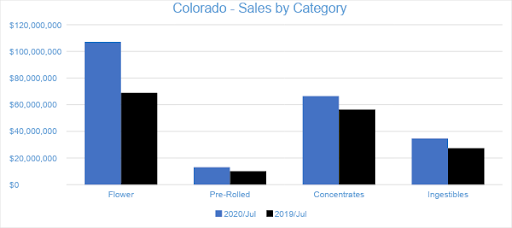
Colorado
July 2020 was another milestone for the Colorado cannabis market. Sales in Colorado’s adult-use and medical dispensaries reached a combined $226.8 million, growing 13 percent from June. Compared to the previous July, sales increased by nearly 35 percent. Adult-use sales grew by 33 percent from July 2019 while medical sales grew by 41 percent. Year-to-date through July, combined channel sales have reached over $1.2 billion, increasing 22 percent from the same period last year.
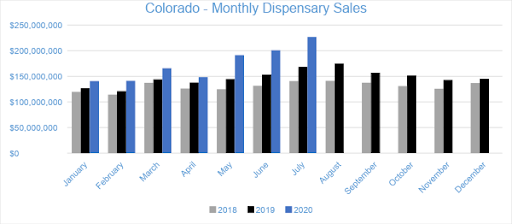
In July, Flower sales accounted for over 47 percent of total monthly revenues, totaling at $107.2 million. Flower sales increased nearly 11 percent from the trailing month and grew 56 percent from the previous July. Year-to-date sales have increased by 40 percent in the category over the same period last year. Pre-rolled Joints, which are tracked separately from Flower, generated $13.2 million in sales for July 2020, growing 31 percent from July 2019 and increasing 17 percent from the trailing month.
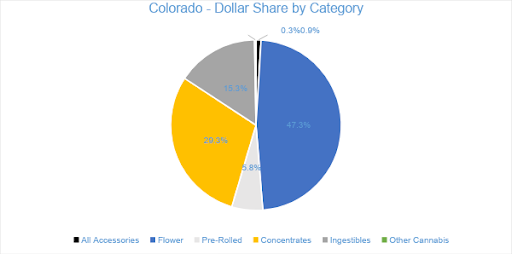
The $66.5 million in Concentrate sales during July 2020 represented an 18 percent increase from the previous year and 15 percent growth from the trailing month. The category accounted for over 29 percent of revenue for the month. Vape products, the largest segment within the Concentrates category, reached $32.9 million in July sales, increasing 19 percent compared to June and four percent from the previous July, respectively. The Vape category accounted almost 50 percent of Concentrates’ revenue and 15 percent of overall sales for the month.
In July 2020, sales from Ingestibles generated $34.7 million, a 27 percent increase from July 2019. Compared to the trailing month, sales in the category grew by 17 percent. Edibles generated $32.7 million in monthly sales revenue, contributing over 94 percent to category sales and 14 percent of total revenue for the month. Sublinguals made up the remaining six percent of Ingestibles sales, totaling $1.9 million for the month.
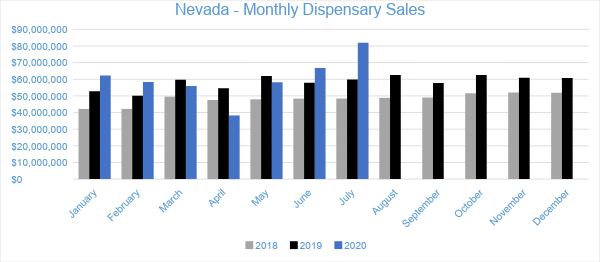
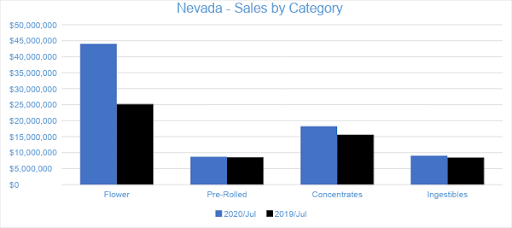
Nevada
In July, Nevada dispensaries generated $82 million in sales. Compared to July of 2019, sales grew by 37 percent and by nearly 23 percent compared to the trailing month. Year-to-date through July, sales have reached $421.9 million, growing by six percent compared to the same period last year.
Flower sales accounted for nearly 54 percent of overall July 2020 sales. Sales in the category reached $44.1 million for the month, growing 27 percent from June. Pre-rolled Joints, tracked separately from Flower, accounted for nearly 11 percent of July sales, with $8.8 million in revenue. Compared to July 2019, Flower and Pre-rolled Joints sales have increased by 75 percent and by two percent, respectively.
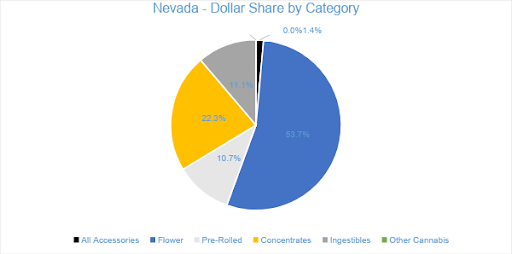
Concentrate sales in July reached $18.3 million, accounting for more than 22 percent of overall revenue for the month. Compared to the trailing month as well as to the previous July, category sales increased by 17 percent. Vape products, the largest segment in the Concentrates category, generated $13.3 million for the month, contributing nearly 73 percent to Concentrates revenue and over 16 percent to total monthly sales.
Sales in the Ingestibles category accounted for 11 percent of all revenues, totaling $9.1 million in July sales and growing by about seven percent from the previous year. Compared to June, category sales grew by 17 percent. Edibles made up 90 percent of the Ingestibles category, reaching $8.2 million in July revenue, and Sublingual sales contributed the remaining ten percent.
Oregon
In July 2020, cannabis sales in Oregon’s medical and adult-use dispensaries reached a combined $106 million. Compared to the trailing month, sales in the Oregon market grew by about six percent, while compared to July 2019, sales grew by 45 percent overall. Year-to-date sales in Oregon have reached $620.3 million, growing by 39 percent compared to the same period in 2019.
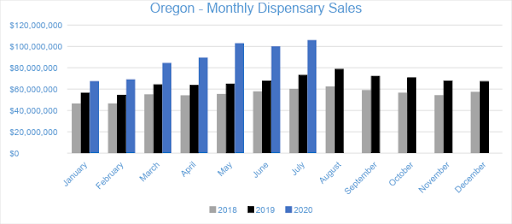
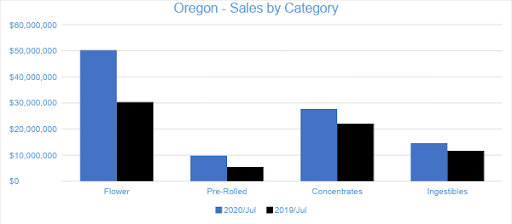
Sales of Flower generated $50.3 million in July 2020, accounting for 47 percent of overall revenues. Compared to the trailing month, Flower sales grew by four percent, and compared to July 2019, sales increased by 65 percent. Pre-rolled Joints, which are tracked as a separate category, accounted for nine percent of overall revenues with $9.8 million in sales.
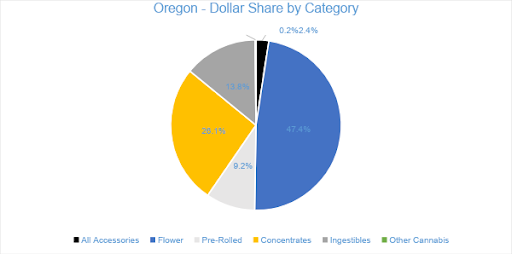
Concentrates generated $27.7 million in July sales. Compared to the trailing month and July 2019, sales in the category increased five percent and just over 25 percent, respectively. Vape products, the largest segment in the Concentrates category, generated $15 million in July, contributing over 54 percent of Concentrates sales and more than 14 percent of total monthly revenue.
In July 2020, Ingestibles generated over $14.6 million in sales, a nine percent increase from the previous month and nearly 25 percent compared to July 2019. Edibles, which made up 83 percent of the Ingestibles category and more than 11 percent of overall monthly sales, generated $12 million in July 2020.
For readers looking for a deeper look at cannabis markets across these five states and more, including segmentation by additional product categories, brand and item detail, longer history, and segmentation by product attributes, learn how the BDSA GreenEdge Platform can provide you with unlimited access to the most accurate and actionable data and analysis.
Source: https://www.newcannabisventures.com/july-cannabis-sales-surge-in-5-western-states/





